The Olson Lab
Drs O'Dell and Olson published a paper that describes a study into the cellular and molecular processes of cerebral cortex development—the brain’s outer layer of neural tissue—is featured on the cover of the July 29 issue of The Journal of Neuroscience, the official publication of the Society of Neuroscience. Reelin Prevents Apical Neurite Retraction during Terminal Translocation and Dendrite Initiation, J Neurosci 35:10659-74, 2015.
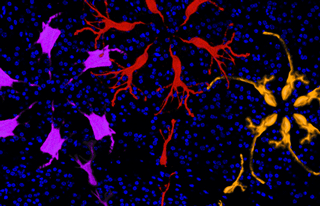
The leading process of a migrating wild-type cortical neuron (red) is shown to bloom explosively into the apical dendrite. In contrast, neurons in reeler cortices (purple, yellow) show normal migrating morphology but their dendritic blooms are duds, with misoriented and simplified arbors. Precise transformation of the leading process may be required for precise migration arrest and proper cortical lamination (shown by Hoechst nuclear staining, blue).
Findings from the NIH-funded study provide insight into how a signaling molecule called Reelin coordinates the growth and wiring of the developing brain and may help explain why patients who have mutations in the RELN gene have profound intellectual disability and epilepsy. The RELN gene provides the code that produce Reelin. Multiphoton live imaging and thereby direct visualization of the embryonic mouse nervous system was used to observe developing neurons in their native environment, both in embryos that included a deficiency of the Reelin molecule and in normal mouse embryos. We provide novel documentation of the dynamic properties of the developing neurite arbor in an ex vivo environment, and elucidate the role of a specific protein (Reelin) in these dynamic interactions during early cortical development. This comparison allowed us to examine the specific differences in cellular dynamics in the two conditions. We discovered that a deficiency in Reelin disrupted the stability of part of the cortical neuron that receives signals from other neurons, a part called the dendrite.

